Introduction
Have you heard about the Schlenk line in class but didn’t quite understand it? Or would you like to set up a Schlenk line in your lab? Whatever the case may be, this article is for you!
First, I’ll explain what a Schlenk line is and how it’s built, then we’ll look at the possible variations. Finally, we’ll go over the main functions of the Schlenk line.
What is Schlenk line? How is it made?
Def. Schlenk Line
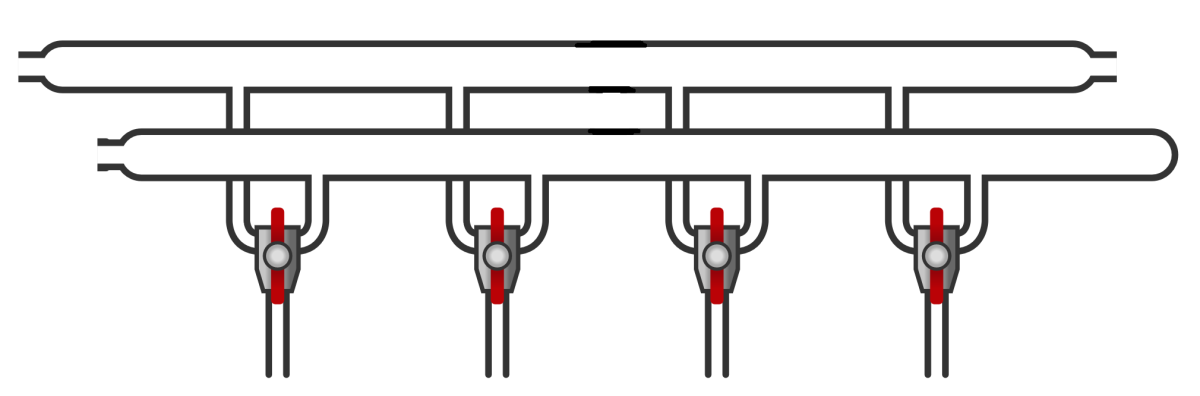
The Schlenk line is a piece of laboratory glassware typically consisting of two glass tubes (also referred to as lines), both connected to various stopcocks at the bottom (Figure 1).
This type of glassware is used to carry out reactions—or more generally, experiments—under a controlled atmosphere.

In an Organic Chemistry laboratory, the two lines of the Schlenk line are typically used as follows:
One line is used to create a vacuum;
The other line serves as a source of inert gas (nitrogen or argon).
Specifically, the vacuum line is useful for removing air from the glassware in which the reaction will take place, while the inert gas line is used to replace the removed air with an inert gas.
All of this can be done by connecting the glassware (reaction flasks, etc.) to the Schlenk line using tubing attached to the outlets of the Schlenk line’s stopcocks.
However, for the Schlenk line to function properly, several additional components are required, such as connection tubing, inert gas cylinders, vacuum pumps, traps, bubblers, and more.
As you can easily imagine, the explanation given so far of the Schlenk line—as a piece of glassware consisting of two lines connected to stopcocks—is certainly not enough to understand how it works and how to use this equipment effectively.
Therefore, it’s necessary to go into more detail. To do this, we’ll first take a closer look at the vacuum line, and then we’ll move on to the inert gas line.
Vacuum line
The vacuum line of the Schlenk line is, as previously mentioned, the glass tube that is typically sealed at one end. This is because the vacuum line must necessarily be a closed system—not open to the air and completely free of leaks.
If the Schlenk line you are using has both glass tubes open at one end, one of them can be sealed off to create a proper vacuum line.
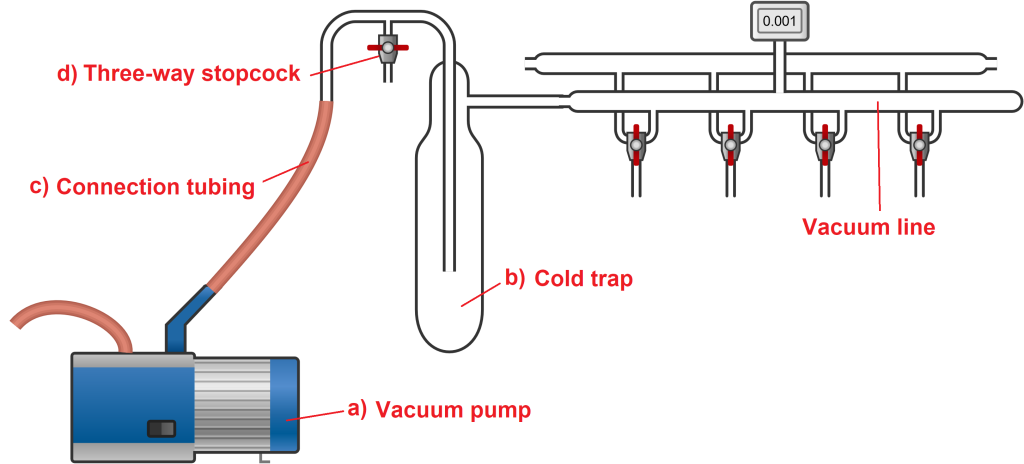
The vacuum line consists of the following essential components:
A vacuum pump, which generates the required vacuum. Typically, oil rotary vane pumps are used, capable of reaching a vacuum down to 0.1 mbar.
A cold trap, installed between the vacuum pump and the Schlenk line, to condense and trap any volatile substances that might originate from the Schlenk line. Without the trap, these vapours could enter and potentially damage the vacuum pump.
Connection tubing, used to connect the pump to the trap and the trap to the Schlenk line.
A three-way stopcock, located between the pump and the trap. This stopcock serves to isolate the vacuum system when the pump is turned on, connecting the pump to the vacuum line. When you are finished using the Schlenk line, the same stopcock allows air to re-enter the line by opening to the atmosphere. In this sense, it functions like an on/off switch: when closed, it maintains the vacuum in the Schlenk line; when open, it lets air back in so that the vacuum pump can be turned off and atmospheric pressure restored.
The diagram below illustrates these four main components of the vacuum line.
Inert gas line
The second glass tube of the Schlenk line can be used as the inert gas line. This line must be open at both ends and is typically used to introduce argon or nitrogen into laboratory glassware.
Naturally, the inert gas line requires a series of essential components to function properly. Below is a possible setup of the necessary accessories, though alternative configurations are also possible.
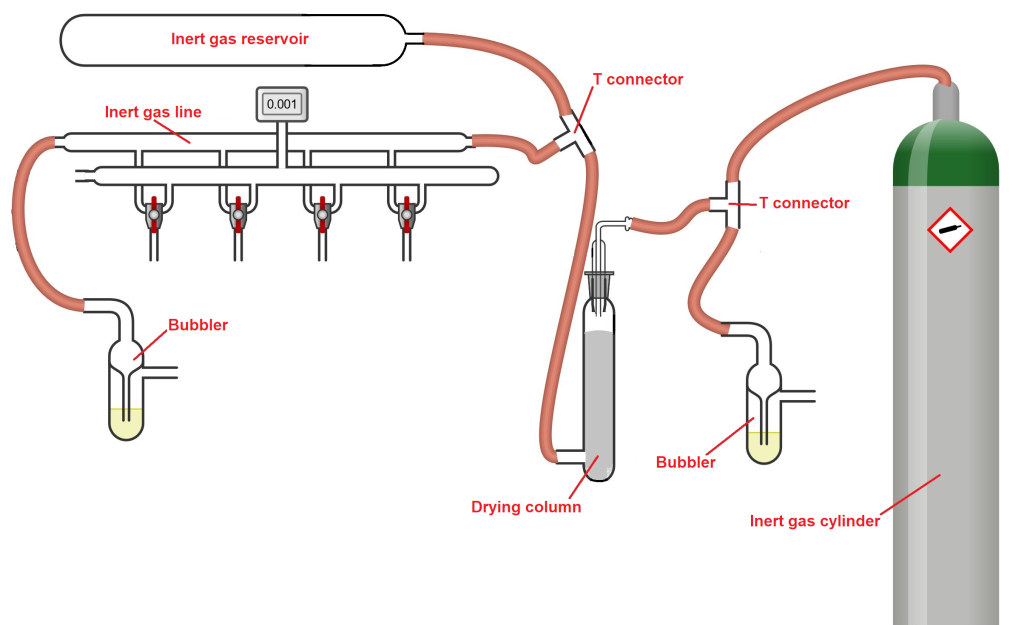
Inert gas cylinder – This is the fundamental source for generating an inert gas flow into the Schlenk line. The cylinder is equipped with a pressure regulator at the top, allowing control over the gas flow rate. The cylinder is connected to the Schlenk line via connection tubing.
Tube connectors – Various connectors may be present in the Schlenk line setup, depending on how many branches or extensions are needed. In the diagram below, for instance, two T-connectors are shown.
Bubblers – These are glass containers filled with a viscous liquid, typically oil or mercury. Inside the bubbler is a glass tube that ends below the surface of the liquid. This tube directs gas from the cylinder into the liquid, forming visible bubbles when the gas flows through. Bubblers serve as visual indicators of gas flow within the Schlenk line. In the figure below, two bubblers are shown: one at the beginning, functioning as a pressure release, in case the drying column becomes blocked; one at the end, indicating that inert gas has successfully reached the Schlenk line. When adjusting the gas flow, you should observe the final bubbler to ensure a steady but gentle bubbling, indicating an appropriate flow rate.
Drying column – This is a glass column filled with a drying agent (commonly P₂O₅) to remove any residual moisture present in the gas or potentially entering from the surrounding environment.
Inert gas reservoir – This is a large glass cylinder that acts as a backup supply of inert gas, useful in case of interruptions or irregularities in gas flow from the main cylinder. While helpful, it is not essential, and not all Schlenk lines are equipped with one.
All of these accessories, as illustrated in the diagram below, are connected to the inert gas line of the Schlenk system using connection tubing.
Variants of the Schlenk Line
As already mentioned, there is not just one Schlenk line, but there can be different variants.
For example, there are Schlenk lines with only one line (a single glass tube); see the figure below.
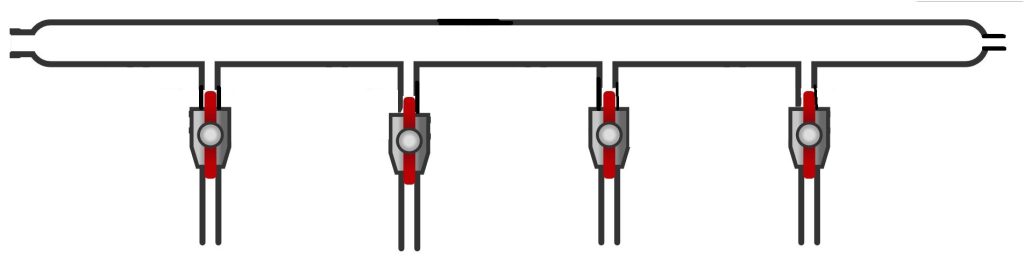
Such Schlenk lines are generally used only as inert gas lines and are often employed to create multiple fixed stations for solvent distillation under an inert atmosphere.
Schlenk Line: How It Works
So far, we have seen how a Schlenk line is built and what its possible variants are. But how does it work?
Let’s consider the most common case in an Organic Chemistry laboratory, namely a vacuum-inert gas Schlenk line.
We previously mentioned that the stopcocks of the Schlenk line are connected both to the vacuum line and to the inert gas line. However, at the terminal end, these stopcocks have a glass joint that serves to connect the valve to the laboratory glassware being used. The connection is made using flexible tubing.
To better understand how to use the Schlenk line, let’s imagine that we need to carry out a reaction under an inert atmosphere. To do this, we will use the vacuum-inert gas Schlenk line in the following way:
1) The reaction flask is connected to the Schlenk line’s connection tube via a glass adapter attached to the flask, or through the stopcock present on the flask itself, in the case of Schlenk flasks (see figure below).
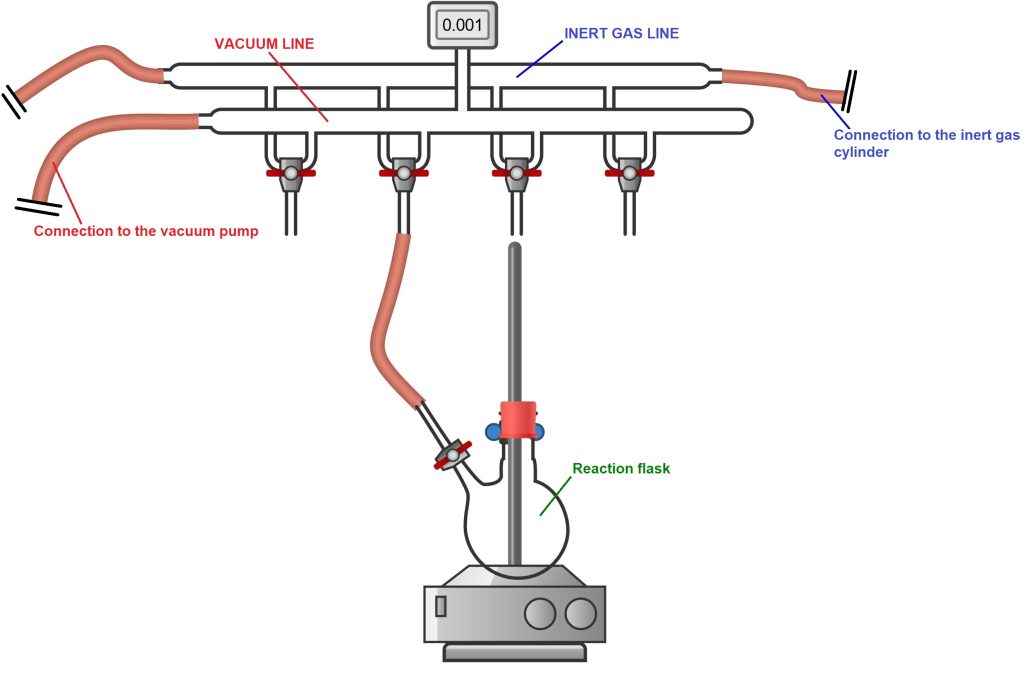
2) The flask is evacuated by opening the stopcock on the flask and turning the Schlenk line stopcock so that only the vacuum line is open (the stopcock is closed to the inert gas line). During this phase, the air inside the flask is drawn out by the vacuum pump. If it is necessary to dry the reaction flask, the flask can be heated with a heat gun. The combined action of heat and vacuum causes any water adsorbed on the glass to detach from the walls and be removed by the vacuum pump.
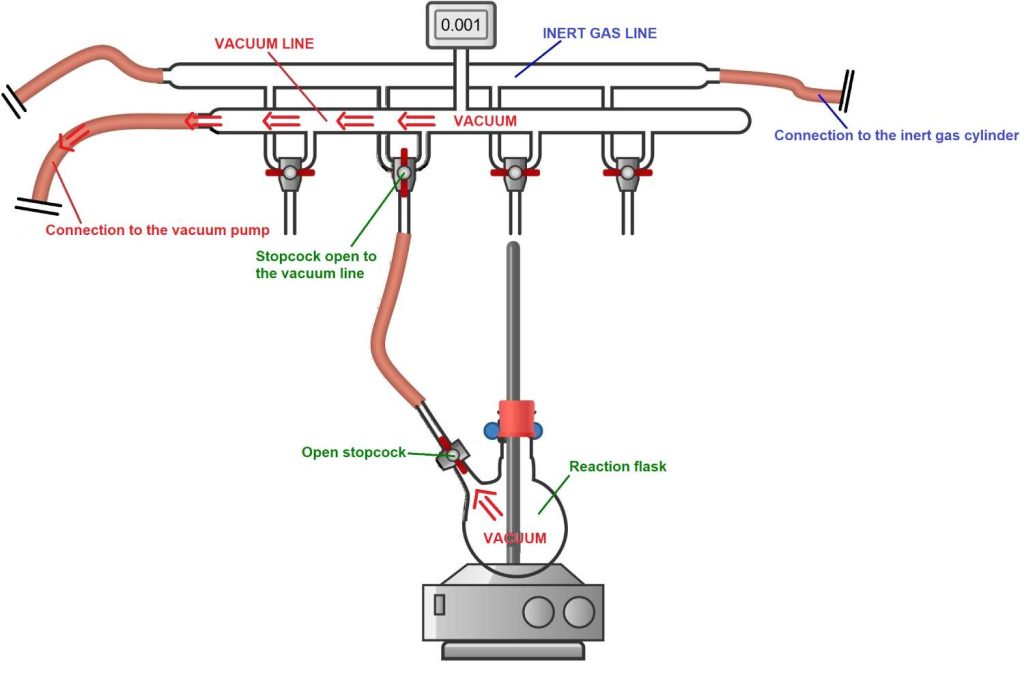
3) After the vacuum phase, the inert gas is introduced into the reaction flask. To do this, simply turn the Schlenk line stopcock toward the inert gas line. In this way, the inert gas fills the flask, replacing the air that was previously removed by the vacuum pump.
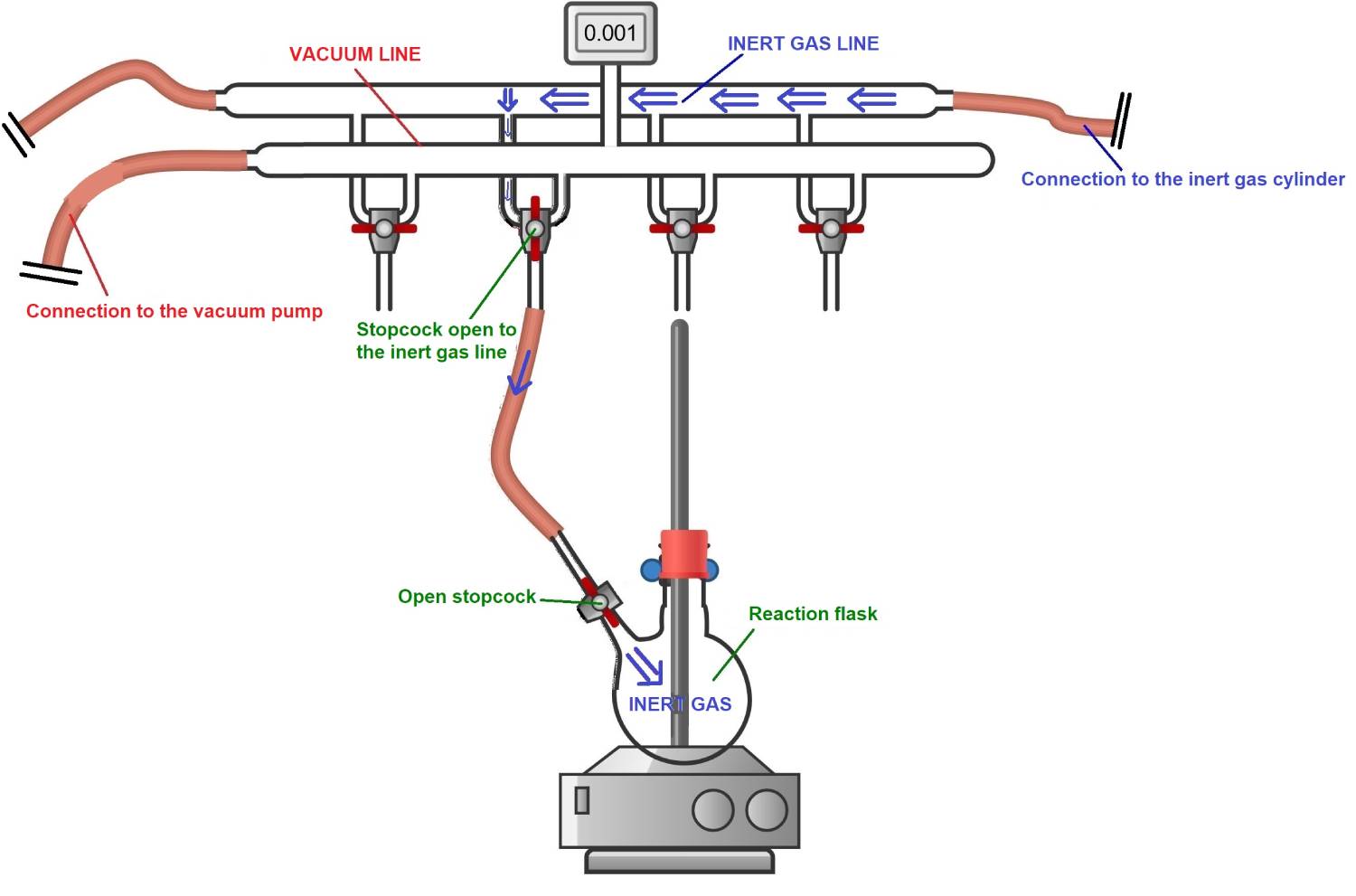
4) The vacuum-inert gas cycle is generally repeated three times in total, along with heating with the heat gun. This ensures the complete removal of moisture and residual air from the reaction flask.
5) Subsequently, it is possible to add the reagents to the flask and start the reaction.
Obviously, other stopcocks on the Schlenk line can be connected to additional reaction flasks or other glassware to perform multiple experiments simultaneously.