
THE UNMISSABLE PUBLICATION
The important publications that a chemist must know. Today we have chosen:
Ullmann e Bielecki – 1901 – Ueber Synthesen in der Biphenylreihe
Ullmann, F.; Bielecki, J. Ueber Synthesen in der Biphenylreihe. Ber. Dtsch. Chem. Ges. 1901, 34 (2), 2174–2185. https://doi.org/10.1002/cber.190103402141.
‘’Ullmann coupling: the first example of transition metal-catalyzed aryl C–C bond formation’’.

Introduction
Ullmann coupling reactions hold a pivotal place in chemistry, as the first example of C-C bond formation facilitated by a transition metal. At the heart of this groundbreaking discovery is copper (Cu), the metal central to Ullmann coupling.
This reaction not only revolutionized organic chemistry but also introduced a transformative concept: the use of transition metal catalysts for C-C bond formation. It is no surprise, then, that we have included the seminal report on the Ullmann homocoupling reaction in our collection of “unmissable publications”.
Ueber Synthesen in der Biphenylreihe
‘Ueber Synthesen in der Biphenylreihe,’ meaning ‘On the Synthesis in the Biphenyl Series’ in German, is Ullmann’s first significant report on the discovery of the homocoupling reaction using copper. [1]
Together with Bielecki, Ullmann reported at the beginning of this article the successful synthesis of 2,2′-dinitrobiphenyl compound with the following reaction:

By heating 1-bromo-2-nitrobenzene at 200°C and gradually adding an excess of copper powder, the homocoupling product (2,2′-dinitrobiphenyl) was obtained with a 76% yield.
Besides 2,2′-dinitrobiphenyl, Ullmann also explored the reaction’s scope with aryls containing different halogen atoms and various substituents. The scheme below illustrates the reaction’s scope.
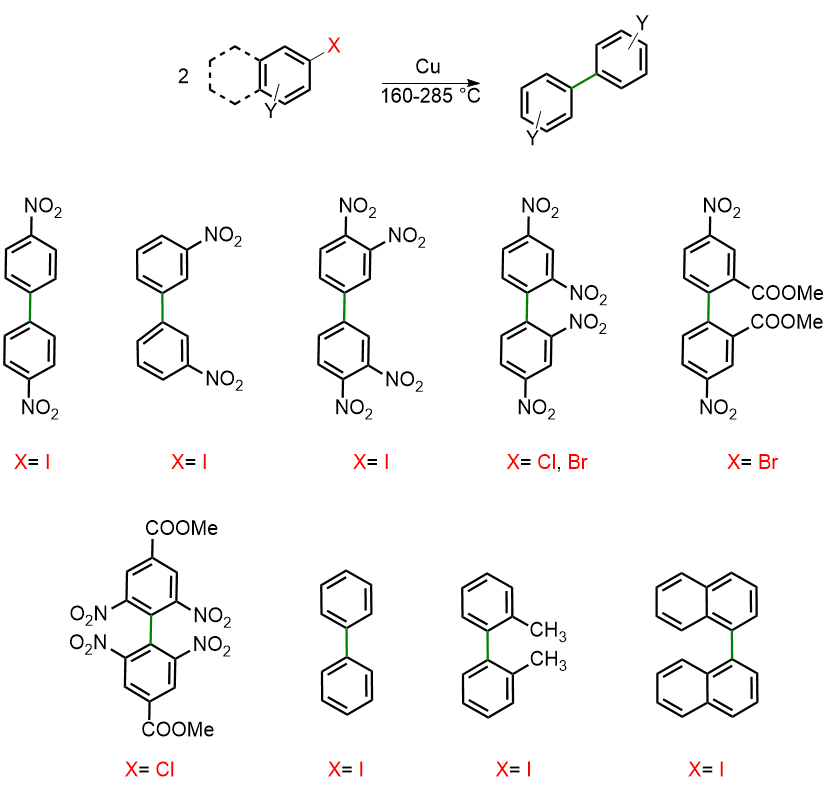
Therefore, Ullmann demonstrated that the reaction can proceed with Cl, Br, and I as halogens. Moreover, the substituent can be either an electron-withdrawing group, such as NO₂ or COOMe, or an electron-donating group, such as -CH₃. The reaction can also occur in the absence of substituents, as shown in the formation of biphenyl and binaphthalene products.
Moreover, Ullmann specified that, in the case of polynitrohalogenated compounds, the reaction becomes more vigorous, necessitating the use of a solvent, such as nitrobenzene, to moderate its intensity.
Conclusion
With this publication, Ullmann showcased to the scientific community the remarkable potential of copper in organic chemistry. For the first time, thanks to Ullmann’s work, this metal demonstrated its ability to facilitate the formation of aromatic C-C bonds.
Since its discovery, the Ullmann coupling has been extensively studied and refined. Today, it is also known as the Ullmann-Ma coupling, thanks to groundbreaking advancements by Ma’s team, who revealed how amine ligands can significantly accelerate the reaction. [2]
References
1) Ullmann, F.; Bielecki, J. Ueber Synthesen in der Biphenylreihe. Ber. Dtsch. Chem. Ges. 1901, 34 (2), 2174–2185. https://doi.org/10.1002/cber.190103402141.
2) Sambiagio, C.; Marsden, S. P.; Blacker, A. J.; McGowan, P. C. Copper Catalysed Ullmann Type Chemistry: From Mechanistic Aspects to Modern Development. Chem. Soc. Rev. 2014, 43 (10), 3525–3550. https://doi.org/10.1039/C3CS60289C.
