Supplies
To perform the red cabbage indicator experiment you need the following products and tools:
Products
Instruments
Time
Water
Red Cabbage
Substances for pH
Knife
Pot
Containers
10 min to boil the red cabbage
20 min to test pH
As you can see from the table, you need a red cabbage, water and all the substances whose pH you want to test. If you want to carry out the experiment better, you can use distilled water instead of tap water. Regarding the instruments, you need what you normally find at home: a pot, a knife and containers such as glasses.
The duration of the experiment depends on how many substances you want to test, in the table above I have put an indicative time of 20 min if you want to test 10-15 substances. Either way, you need at least ten minutes to boil the cabbage.
Instructions
The operations to do are very simple:
- Cut the red cabbage into small pieces;
- Put the chopped cabbage in a pot with a little water;
- Boil everything for 10 minutes;
- Filter with a strainer to obtain the liquid part;
After that, you will have obtained a purple liquid, which is our cabbage indicator. With this liquid, you will be able to measure the pH of all substances that come your way. Just add a bit of indicator (2-3 mL) to the solutions you want to test. By observing the colour change, you can find the pH.
Background of the experiment
pH Scale
This cabbage extract has the ability to change colour based on the pH of the solution it is in.
pH is a way for chemists to communicate with each other and tell each other whether a solution is acidic or basic. An acidic pH ranges from 0 to 6, a neutral pH is 7, a basic pH ranges from 8 to 14; where 0 is the maximum acidity and 14 is the maximum basicity. Our cabbage extract helps us derive these pH numbers by communicating with us via colour. In fact, cabbage associates a different colour for each pH level, for example red is a pH of 0, blue is a pH of 7, therefore a neutral pH, and yellow is a pH of 14 and so on for the intermediate colours. By simply looking at the scale in Figure 1, you will now know how to measure pH like a real chemist and can calculate the pH of any substance that comes your way.

Acids and bases – quick explanation
If you don’t know what an acid or a base is, don’t worry, we can see it in two lines.
An acid is a substance that releases H+ ions, positively charged particles, into solution.
While a base is a substance that releases OH- ions, ions with a negative charge; for neutral, on the other hand, we mean a solution where the quantity of H+ and OH- ions is equal.
Experiment
In the experiment we performed and which you can see in the video above, we tested the pH of 10 substances and calculated the pH simply by comparing the colour obtained after adding the cabbage extract with the corresponding number on the pH scale in Figure 1. The tested substances are the following:
- Sulphuric acid. The solution, prepared by adding a bit of acid to water, turned red when the indicator was added, indicating a pH of 0, i.e. very acidic.
- Hydrochloric acid, HCl. The solution turned red again indicating a pH around 0, therefore acidic.
- Lemon. We squeezed some lemon into the water and upon adding the indicator, the solution turned salmon colored indicating a pH around 1-2.
- Sugar. After dissolving a teaspoon of sugar in water and adding the indicator, the solution turned steel blue, indicating a neutral pH, i.e. equal to 7.
- Baking soda. After dissolving a teaspoon of sodium bicarbonate in water and adding the indicator, the solution turned cadet blue, indicating a pH of around 8.
- Dish soap. After dissolving some dish soap in water and adding cabbage extract, the solution turned light purple, thus slightly acidic pH around 6.
- Bleach. Upon addition of the extract, the bleach turned yellow-green, showing a pH of 12-13.
- NaOH. After dissolving a teaspoon of caustic soda in water and adding the indicator, the solution changed from colourless to yellow, indicating a pH of 14, therefore very basic.
- Potassium carbonate. This salt was dissolved in water and after adding the extract, the solution took on a beautiful green colour, indicative of a pH of 10, therefore basic. Note that carbonate is much more basic than the bicarbonate analogue tested in step 5.
- Schweppes soft drink. We added the indicator to the drink and the solution turned purple-red indicating a pH between 1 and 2, therefore quite acidic..
The picture below (Figure 2) summarizes our complete experiment.
If you want to know how cabbage changes colour based on pH, read the next paragraph.

Scientific explanation
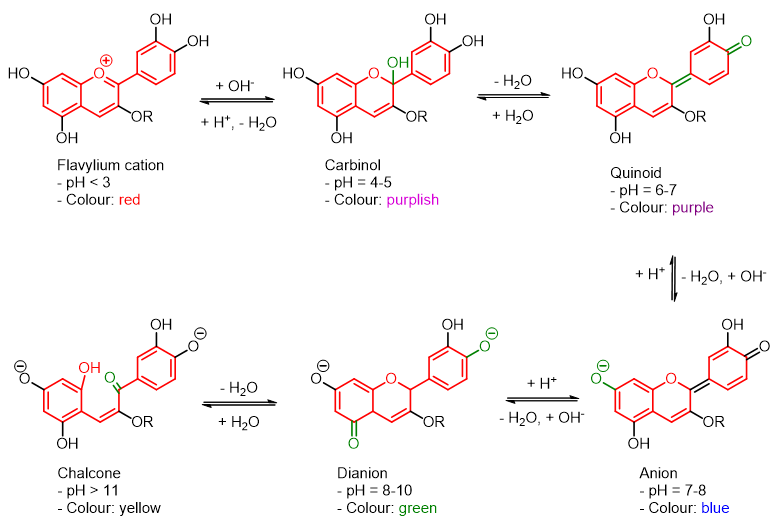
How does cabbage actually change colour? This happens thanks to the special substances that cabbage contains, these substances are known as anthocyanins, a word that derives from the Greek anthos (flower) and kyanos (blue). Anthocyanins are a class of natural substances that belong to the larger family of flavonoids. In nature, there are at least 500 different types of anthocyanins, all sharing a similar chemical structure: an anthocyanidin to which various sugar molecules bind. Anthocyanidin is the basic building block of all existing anthocyanins. There are only 23 anthocyanidins and they are all characterized by the flavylium cation, which you can see in Figure 3. Depending on the R substituents present on the flavylium cation, the various types of anthocyanidins can be obtained. The 500 different types of anthocyanins are instead created by the position and type of sugar that binds to the flavylium ion. Typically sugar molecules bind at the R3 or R5 position.

However, to explain the behaviour of red cabbage, we need only one type of anthocyanidin, the one most abundant in this food: CYANIDIN, whose structure is shown below.

It is precisely this molecule to interact with the H+ and OH- ions present in the surrounding environment and is therefore sensitive to pH.
In fact, as you can see from the scheme below, the molecule takes various shapes based on the pH. These different structures absorb light differently, thus causing the typical colour change of the cabbage extract as observed in our experiments
This lesson

The cabbage experiment
€
1.50
Download as pdf (unchageable) file

The cabbage experiment
€
2.20
Downoald as docx (editable) file



