Supplies
To perform the egg in vinegar experiment you need the following substances and instruments:
| Products | Instruments | Time |
| Egg | Glass/Container | 1-2 days |
| Vinegar |
As you can see from the table, you need a fresh egg, vinegar, preferably white wine one, a glass or similar container and a bit of patience, since it takes at least a few days to see the desired effect.
Instructions
The instructions to follow are very simple:
- Fill the glass with vinegar
- Insert the egg inside
- Leave the egg in the vinegar for several hours
- Observe
Observations
You will immediately start to observe bubbles coming up from the vinegar and over time (8-10 hours) you will notice how the shell is slowly consumed by the vinegar. When the shell consumption reaction is complete, you can pull the egg out of the vinegar and you can notice how the consistency of the egg has changed. That is a new rubbery and bouncy egg.
Scientific explanation
However, what happened to the egg? Let’s see the complete scientific explanation.
The shell of the egg is made up of calcium carbonate (CaCO3), while the vinegar has a certain percentage of acetic acid (CH3COOH) in it. These two substances, calcium carbonate and acetic acid react together with a reaction called acid-base. The acid-base reaction takes place between an acid, in our case acetic acid, and a base, in our case calcium carbonate. You can see the reaction in more detail in the image below.
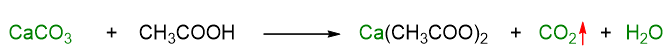
Looking at the reaction, you can observe how all the calcium carbonate, in green, is converted into carbonic acid (H2CO3), which, however, is unstable and immediately transforms into carbon dioxide, CO2, and water, H2O. The formed carbon dioxide is actually a gas and will tend to escape from the glass giving rise to the bubbles that we have noticed forming since the beginning of the experiment (Figure 2).
As you can also see from the reaction, another substance is formed in the products: calcium acetate Ca(CH3COOH)2, which, however, is soluble in water and indeed you can’t notice it in the solution where the egg is.
In other words, we can summarize that the acetic acid gradually eats the shell of the egg, turning it into gas and water

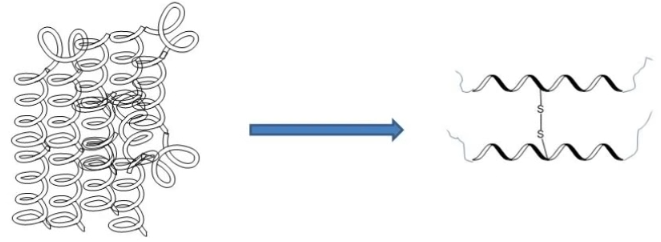
That is not all though. Once the shell has fallen apart, the acetic acid goes on with its destructive action and this time it picks on the egg white. In fact, it reacts with the proteins in the egg white, forcing them to change shape and form disulfide bridges. These bridges consist of the union of two sulfur atoms, which are present within proteins; the disulfide bridges give elasticity to the protein chains and therefore the egg white changes consistency (Figure 3). It will no longer be runny, like in raw egg, but will become chewy and bouncy.
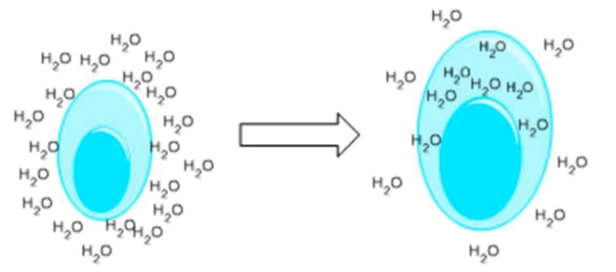
Finally, you will notice that the egg has also become bigger; this is because it has absorbed water. In fact, due to the phenomenon called osmosis, the water present in the vinegar will tend to enter the egg, which by now is shell-less. Osmosis is nothing more than the passage of a liquid from an area where it is more present to an area where it is less present. Consequently, since there is much more water in the glass than inside the egg, the water will tend to enter the egg and swell it (Figure 4).
Here is how to have a bouncy and swollen egg thanks to a bit of vinegar. Now you can also understand why some chefs add a few drops of vinegar to the boiling water in which they make hard-boiled eggs: this will make it easier to remove the shell.
This article

Egg in vinegar
€
1.50
Download pdf (unchangeable file)

Egg in vinegar
€
2.20
Download docx (changeable file)


