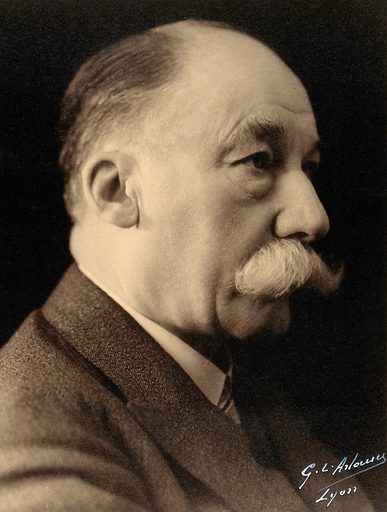The unmissable publication
The important publications that a chemist must know. Today we have chosen:
GRIGNARD, V., Compl. rend., 130, 1322 (1900).
‘’Sur quelques nouvelles combinaisons organométalliques du magnésium et leur application à, des synthèses d’alcools et d’hydrocarbures’’.

INTRODUCTION
On May 14, 1900, the chemist Henri Moissan presented to the Académie des Sciences in Paris a rather brief report entitled ” Sur quelques nouvelles combinaisons organométalliques du magnésium et leur application à, des synthèses d’alcools et d’hydrocarbures ” (About some new organometallic combinations of magnesium and their application to the synthesis of alcohols and hydrocarbons). This report was later published in the journal Comptes Rendus and became the publication that revolutionized the world of chemistry. The author of this article was the man who hated chemistry: Victor Grignard and this was his first publication in this field.
Before summarizing the content of the article, let’s briefly see who was this chemist who hated chemistry and the state of the art before his publication.
V. GRIGNARD: THE LIFE
‘’Victor Grignard. Photograph by GL Arlomer, Lyons’’ by Wellcome Collection [CC BY 4.0] via https://www.lookandlearn.com…
Franqois Auguste Victor Grignard was born on May 6, 1871 in Cherbourg from a modest family. His father Theophyile Henri Grignard was a sailmaker and his mother was Marine Arsenal. His parents had to make quite a few financial efforts to allow their son a good education. However, Grignard did not fail to repay the trust of his parents by obtaining excellent results from the beginning of his studies, especially in mathematics.

Would you like to print this article or save it to your computer and read it offline? You can download the pdf file here. Alternatively you can also find the docx file if you need to make changes to the document.
He first attended the local school (1883-1887) and then the École Normale Spéciale in Cluny and the University of Lyon. He graduated with a degree in mathematics in 1894. His education clearly indicated that Grignard would become a mathematics teacher. However, that didn’t happen.
In fact, although Grignard had never thought of approaching chemistry and indeed hated it considering it an inferior, mnemonic and purely empirical discipline, one of his classmates at Cluny stunned him one day. He told him that his vision of chemistry would certainly change if he entered a laboratory. Thus, it was that Grignard accepted an assistant position in a laboratory at the University of Lyon.
Here the chemist Louis Bouveault introduced Grignard to laboratory techniques and in a few weeks the idiosyncrasy for chemistry turned into pure passion. Grignard became enthusiastic about laboratory life and continued to work tirelessly.
He then became a student of the chemist Philippe Antoine Barbier, the head of the department. It was Barbier himself who gave Grignard an old project of his that had ended up in the background. The project concerned organo-magnesium and was entrusted to Grignard for his thesis work.
This work made Grignard famous since its first publication in 1900. In fact, even before his thesis came out, a presentation had already been requested by Moissan in Paris; however Grignard refused and preferred to finish the job by dedicating himself exclusively to his university. On July 18, 1901, Grignard obtained the title of Doctor of Physical Sciences in Lyon with a thesis entitled: ” Sur les combinaisons organomagnésiennes mixtes et leur application à des synthéses d’acides, d’alcools et, d’hydrocarbures ” (On mixed combinations of organomagnesium and their application to the synthesis of acids, alcohols and hydrocarbons). After this work Grignard became world famous and the term Grignard reagents came into common use.
In 1912 his work on organomagnesium was awarded the most prestigious of prizes: the Nobel Prize. [1]
STATE OF THE ART AT THE TIME OF GRIGNARD
Let us now go over what was known to chemists before the publication of Grignard, the subject of this article.
The first synthesized organometals compounds were organolithium, RLi, and organosodium, RNa. However, these compounds were of little practical interest because they were highly flammable. Organomercuries, on the other hand, were also already known, but because they were too stable, they were not very reactive. Therefore, prior to Grignard’s discovery, the only organometals that looked most promising were organozinc, R2Zn. However, these compounds discovered by Frankland in 1849, [2] spontaneously ignited in air and were, hence, difficult to handle. Furthermore, some reactions with such compounds gave unsatisfactory yields and the reactions often took weeks or months to occur.
Therefore, it was clear that an alternative for the organozinc was needed. It was then that in Barbier’s group, which also included Grignard, magnesium was thought like a possible alternative. In fact, this metal has a certain analogy with Zn, but has a greater reactivity.
The first studies that were done on organomagnesium date back to 1891-1894 in the laboratory of Lothar Meyer.[3] Here the symmetric organomagnesiums, R2Mg, were studied and it was discovered that these compounds were not liquids, like those with zinc, but were solids insoluble in most solvents. They also ignited spontaneously in air and in the presence of carbon dioxide.
Given the characteristics, organomagnesium seemed destined to end up in oblivion. However, in 1898 Barbier, Grignard’s teacher, tried to apply Saytzeff’s method, hitherto used for zinc, for magnesium. This method consisted in mixing the two compounds, to be reacted together, with the magnesium, thus avoiding the synthesis of the organometal. Barbier tried this technique with magnesium in the following reaction: [4]

Barbier did not report the yield of this reaction, but stated that such a reaction was not possible with organozincs. He continued the study of magnesium in similar reactions, but the results were so erratic that he decided to put the project on hold.
However, when Grignard was looking for a topic for his doctoral thesis, Barbier proposed organomagnesiums in order to continue the interrupted study. The first results of this study, undertaken by the young Grignard, were published in the work that is the subject of this article.
GRIGNARD, V., Compl. rend., 130, 1322 (1900).
Grignard in this work on Comptes rendus of 1900, which was his third publication as a chemist, describes his new approach for organomagnesiums.
In fact, while trying the Barbier’s method, described above, Grignard faced the same problems of his master, so he decided to change the approach.
In fact, he thought of going back to synthesizing organomagnesium, but this time using the method that Frankland had used for organozinc. This method consisted in heating the alkyl halide in ether in the presence of zinc. [5]
Grignard found that magnesium reacts similarly: in the presence of ether it attacks alkyl halides at ambient T and ordinary P to give a compound which is completely soluble in ether.
He formulated the reaction and structure of organomagnesium as follows:

Unlike the organozincs, the synthesized organomagnesiums do not ignite in the air.
Grignard, in this article, also explained that organomagnesiums can then be used for subsequent reactions with electrophiles. Indeed, he proposed the following example:

In this article, he therefore described the preparation of a certain number of secondary alcohols, starting from aldehydes, and tertiary alcohols, starting from ketones. He stated that the reaction gave excellent results with aliphatic alkyl halides. While, benzyl halides gave a certain amount of unwanted product, such as dibenzyls.
Here are some examples of products prepared by Grignard using organomagnesiums:

Grignard did not report the yields of these reactions. He also added that with α-β unsaturated aldehydes and ketones, it was easier to obtain dehydration from alcohols to give dienes. He proposed the following example:

Grignard concluded the brief publication, we have described here, saying that he was busy finding further applications for the new organometals.
And so it was. His further discoveries were published in his Thesis and in the hundreds subsequent articles that Grignard dedicated to the same topic.
We decided to summarize Grignard’s first article on organomagnesium because it was from that article that everything began and a new road for organic synthesis was inaugurated.
With this article, Grignard demonstrated to the academic world how it was possible to change the reactivity of the carbon atom, from electrophile, in R-X, to stable nucleophile, in RMgX. An important discovery to create new C-C bonds.
References
1) Rheinholdt, H. (1950). Fifty years of the Grignard reaction. Journal of Chemical Education, 27(9), 476. https://doi.org/10.1021/ed027p476;
2) Frankland, E. (1877). Experimental researches in pure, applied, and physical chemistry. In John Van Voorst eBooks. https://scholar.google.com/;
3) Löhr,P. (1891). “Inaug. Dissertation”, Tübingen, 1889; Ann., 261, 48;
Fleck, H. (1893) “Inaug. Dissertation,” Tübingen, 1892; Ann., 276, 129;
Waga. F. (1894) “Inaug. Dissertation.” Tubinxen. 1894: Ann., 282, 320
4) Barbier, P. A. (1899) Compt. rend., 128, 110;
5) Frankland, E. (1859) Ann., 111, 63.
This lesson

Grignard-1900
€
1.50
Download as pdf (unchageable) file

Grignard-1900
€
3.00
Downoald as docx (editable) file