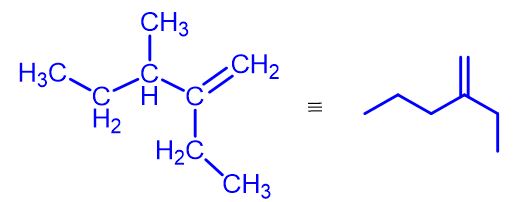
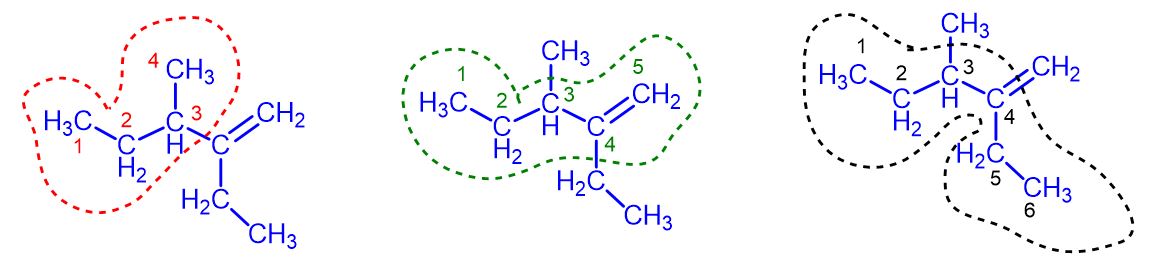
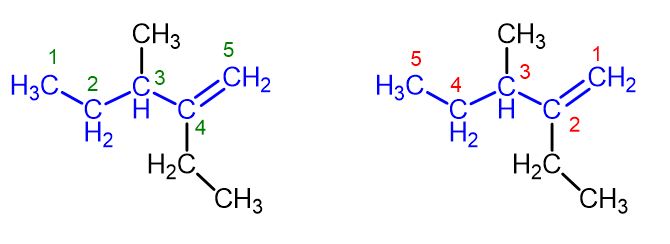
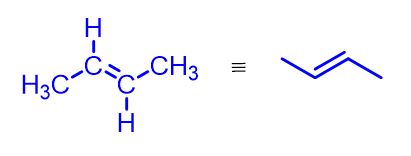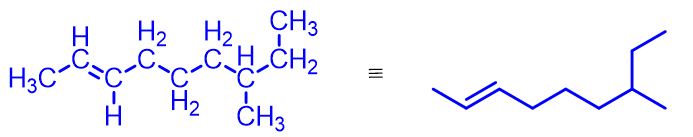Introduction
Are you at the beginning of your Organic Chemistry studies and finding alkenes difficult to understand? Then this article is for you! In fact, with simple words and no unnecessary talk, this seemingly confusing topic will quickly become clear. Moreover, since this site was created to support students, you will have access to the following tools as additional help:
The PDF or DOCX file of this article, so you can review the notes offline;
A free quiz to test your knowledge after reading the article;
A downloadable concept map to get a visual summary of the entire topic.
And now it’s time to get started! Arm yourself with a little patience and confidence in your abilities. Happy reading!
What are alkenes?
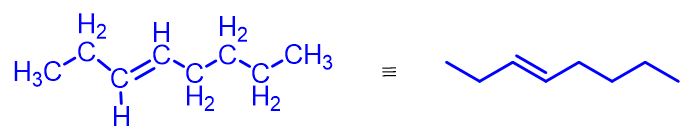
Alkenes are organic compounds made up of only carbon (C) and hydrogen (H). Their general formula is CnH2n, where n is an integer greater than or equal to 2.
So, the first alkene has n = 2 and the general formula C2H4, commonly called ethylene, although the IUPAC name is ethene. Next comes propylene (or propene) with the formula C3H6, but-1-ene with C4H8, pent-1-ene with C5H10, and so on. The figure below illustrates these first four simplest alkenes.

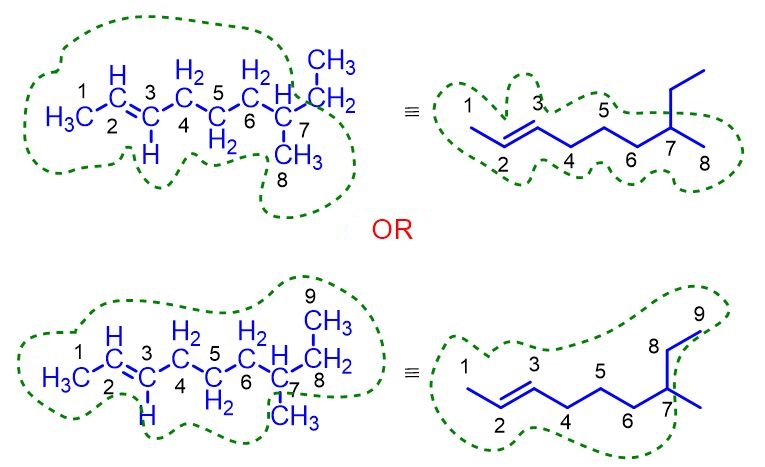
Alkene nomenclature
Alkene nomenclature is not very different from that of alkanes. In fact, the first part of their name, the root, is identical to that of alkanes and indicates the total number of carbon atoms. For 2 carbon atoms the root is eth-, for 3 prop-, for 4 but-, while starting from 5 carbon atoms the root derives from the Greek numerical prefix. The table below lists all the roots from 2 to 20.
Alkene nomenclature, however, differs from that of alkanes for three reasons:

Are you sure you’ve understood how to name alkenes? If you’re still confused, I’ve provided some solved exercises below that put the theory into practice. If, on the other hand, you think you’ve understood everything, try the exercises on your own.
Solved Exercises
Note: If you have any doubts about the nomenclature of more complex organic molecules, check out our comprehensive article, “All Organic Chemistry Nomenclature,” which covers organic nomenclature in its entirety and provides a complete and definitive overview of the topic.
Traditional Nomenclature
Some alkenes still retain a nomenclature different from the IUPAC system, known as “traditional nomenclature,” because it originated from the common language used by scientists before the development of a systematic and universal naming system.
However, traditional nomenclature is still widely used, so it is important to learn it in order to communicate effectively with chemists—especially the more senior ones!
In the table below, you will find the most commonly used traditional names for alkenes, alongside their corresponding IUPAC names.
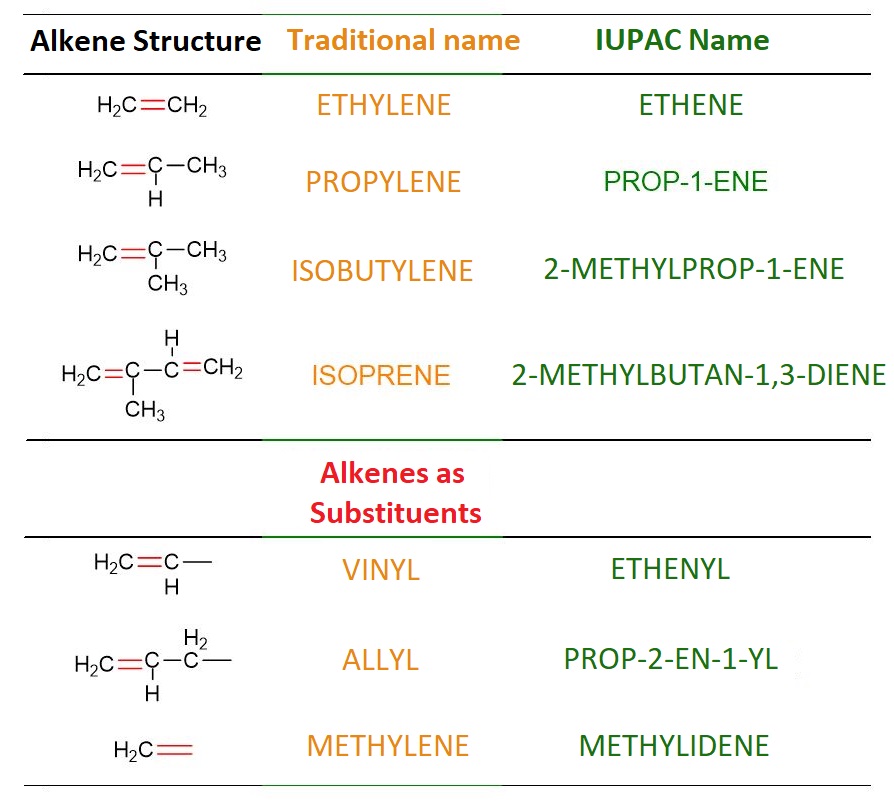
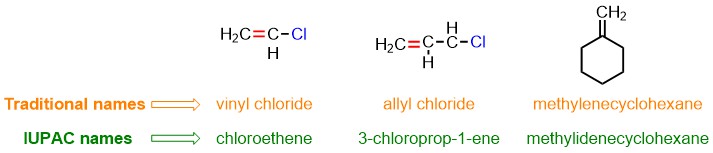
In the last three rows of Table 2, some examples of traditional names for substituent alkenes are reported. These names can be used to designate certain olefins, as shown in the figure below.
Alkene hybridization
In the article on alkanes, we learned that the carbon atoms in these molecules are all sp³ hybridized.
But what is the hybridization of alkenes?
In fact, all the carbon atoms in an alkene that are NOT involved in the double bond will continue to have sp³ hybridization and therefore a tetrahedral geometry.
The hybridization changes only for the carbon atoms involved in the double bond. These, in fact, will have sp² hybridization.
But what does this mean? Let’s recall the electron configuration of carbon. It has two electrons in the 2s orbital and two electrons in the 2p orbitals. However, in order for the carbon atom to form four bonds, one of the electrons in the 2s orbital is promoted to an empty 2p orbital. See the animated figure below.

At this point, there are four unpaired electrons in the s and p orbitals. To form the double bond, these orbitals undergo hybridization—that is, they mix together to create a new set of orbitals capable of forming the double bond. Unlike sp³ hybridization, where all four orbitals mix together, in sp² hybridization only three orbitals hybridize (one s orbital and two p orbitals, px and py), while the remaining orbital (the pz orbital) remains unchanged.
When these three orbitals hybridize, they change shape to adopt a new form that reflects the characteristics of all three original orbitals. Since the s orbital is spherical and the p orbitals are dumbbell-shaped (bilobed), the resulting sp² hybrid orbitals have a shape that is intermediate between spherical and bilobed. See the sp² orbitals in the image below.
These hybridized orbitals adopt a planar arrangement in space, with 120° angles between one orbital and another.
The pz orbital, which did not hybridize with the others, remains unchanged and lies above and below the plane of the sp² hybrid orbitals (last image in Figure 4).

But how does the double bond actually form?
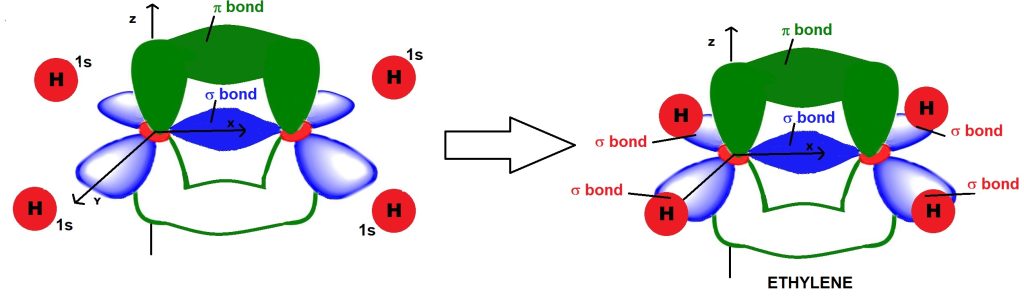
Let’s make two sp² hybridized carbon atoms interact with each other. One sp² orbital of one carbon atom interacts with an sp² orbital of the other carbon atom to form a σ-type bonding orbital (shown in blue in Figure 5).
The σ orbital has cylindrical symmetry with respect to the axis connecting the two carbon nuclei. In other words, this orbital resembles a sort of “sausage-shaped” region extending around the imaginary axis passing through the two interacting nuclei.
However, both carbon atoms also possess an unhybridized pz orbital. The unhybridized pz orbitals interact laterally with each other, creating a π-type bonding molecular orbital, with electron density located above and below the xy plane.

The π orbital is in fact the orbital responsible for the formation of the double bond in alkenes. Compared to a σ orbital, it is weaker because it has a node along the axis passing between the two interacting nuclei and originates from two orbitals that overlap only laterally and only at two regions.
However, the double bond between two carbon atoms is overall stronger than a single C–C bond, because the two nuclei are held together by both a σ bond and a π bond, whereas a single C–C bond consists of only one σ bond.
What happens to the other two sp² hybrid orbitals present on each carbon? These orbitals can be used to form additional σ bonds with other carbon atoms or with hydrogen atoms.
For example, in ethene (C₂H₄), the sp² hybrid orbitals that are not involved in forming the double bond each interact with an s orbital of a hydrogen atom through σ interactions (Figure 6).
Bond lenght
The C=C double bond in alkenes is shorter than the C–C single bond in alkanes. This is because, in the double bond, the combined presence of the π bond and the σ bond adds electron density between the two carbon nuclei, compared to the electron density provided by the σ bond alone in alkanes.
If we take the ethylene molecule as an example again, we see that the C=C bond length is 1.34 Å (Figure 7), whereas the C–C bond length in the corresponding alkane (ethane) is 1.54 Å. However, if we also compare the C–H bond lengths in ethylene and ethane, we notice that these bonds are also shorter in ethylene (1.09 Å vs 1.10 Å).
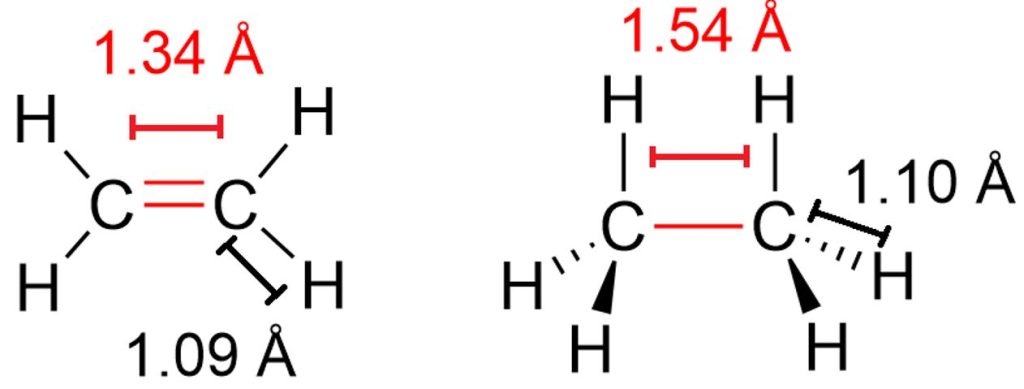
The shortening of the σ bonds of the two carbon atoms involved in the double bond is a consistent feature in alkenes and is due to the sp² hybridization of these atoms. In fact, compared to sp³ hybridization, sp² hybridization involves a greater contribution of the s orbital to the resulting hybrid orbitals, since one s orbital and two p orbitals interact. By contrast, in sp³ hybridization the contribution of the s orbital is lower, because the interaction involves one s orbital and three p orbitals. Since the s orbital has electron density very close to the nucleus, the greater s character of the sp² hybrid orbitals makes them more concentrated around the nuclei than sp³ orbitals. As a result, all σ bonds formed by an sp²-hybridized carbon atom are shorter.
The bond lengths for the first four alkenes are reported in Table 3 below. Generally, the typical length of an (sp²)C=C(sp²) bond is around 1.34–1.35 Å, whereas that of an (sp³)C–C(sp³) bond is about 1.54–1.57 Å. Slightly shorter are the distances for an (sp²)=C–C(sp³) bond, with values ranging between 1.48 and 1.51 Å..[1-5]

cis/trans isomerism
The presence of a double bond in alkenes gives rise to a particular type of isomerism that is not found in simple alkanes. This type of isomerism is known as geometric isomerism and involves distinguishing between two isomers: the cis isomer and the trans isomer.
In particular, the presence of the double bond makes the molecule rigid at that point, meaning it is unable to rotate. While there is free rotation around a single C–C bond at room temperature, rotation around a C=C bond is prevented by the presence of the additional π bond. For a C=C bond to rotate, the π bond would need to be broken, which can occur only at very high temperatures (around 300°C), under acidic conditions, or through photocatalysis. It is precisely this rigidity of the C=C bond that makes cis–trans isomerism possible.
But what are these cis and trans isomers? Let’s look at an example.
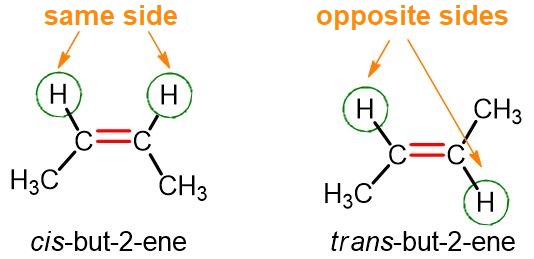
Let us consider the first alkene that exhibits this type of isomerism, namely but-2-ene. From the image below, it can be seen that in the cis isomer of but-2-ene the two hydrogen atoms are on the same side, and the same is true for the two CH₃ groups. In the trans isomer, on the other hand, the two hydrogen atoms are on opposite sides, as are the two CH₃ groups.
In the case of disubstituted alkenes, such as but-2-ene (that is, with one hydrogen and one substituent on each carbon involved in the double bond), we can say that:
The cis (or Z) isomer is the one in which the two hydrogen atoms (or the two substituents) are on the same side;
The trans (or E) isomer is the one in which the two hydrogen atoms (or the two substituents) are on opposite sides.
Not all alkenes exhibit this type of isomerism. More specifically, alkenes:
Monosubstituted alkenes never exhibit cis–trans isomerism;
Tri- and tetrasubstituted alkenes exhibit cis–trans isomerism in almost all cases, except when there are two identical substituents on the same carbon atom involved in the double bond.
The image below illustrates these concepts with practical examples. It is worth noting that for tri- and tetrasubstituted alkenes, the cis/trans nomenclature is difficult to apply; therefore, the E/Z system is preferred, which has specific rules for assigning the two isomers. We will not cover these rules in this article, but I will soon provide a dedicated article on this topic.
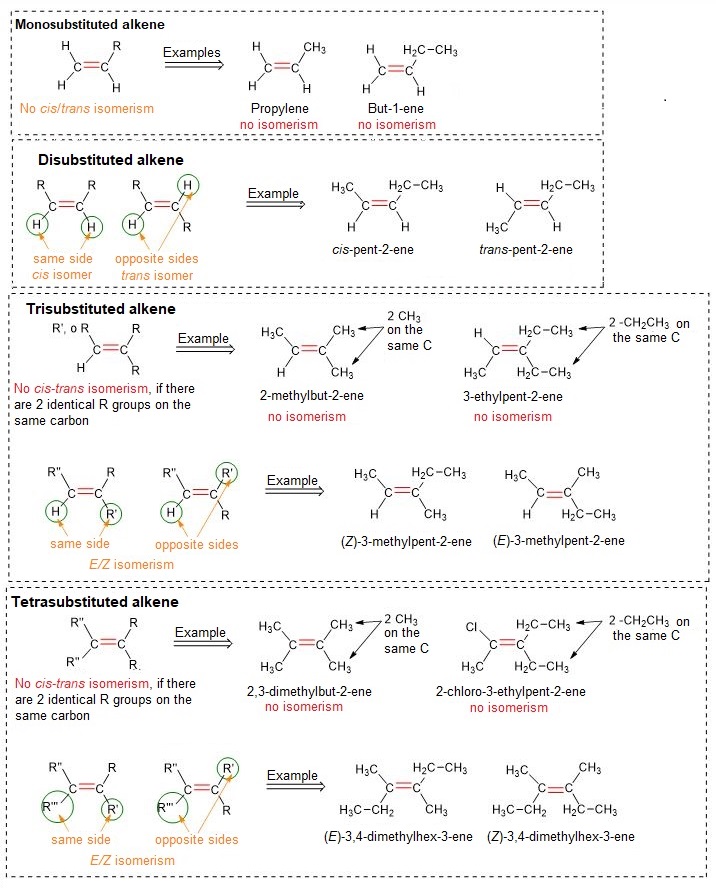
Stability of alkenes
The image below shows a general stability scale of alkenes. The following information can be observed:
Tetrasubstituted alkenes are more stable than trisubstituted ones;
Trisubstituted alkenes are more stable than disubstituted ones;
Among disubstituted alkenes, the trans isomer is always more stable than the cis isomer;
Disubstituted alkenes are more stable than monosubstituted ones;
Monosubstituted alkenes are more stable than ethylene (that is, a non-alkylated alkene).
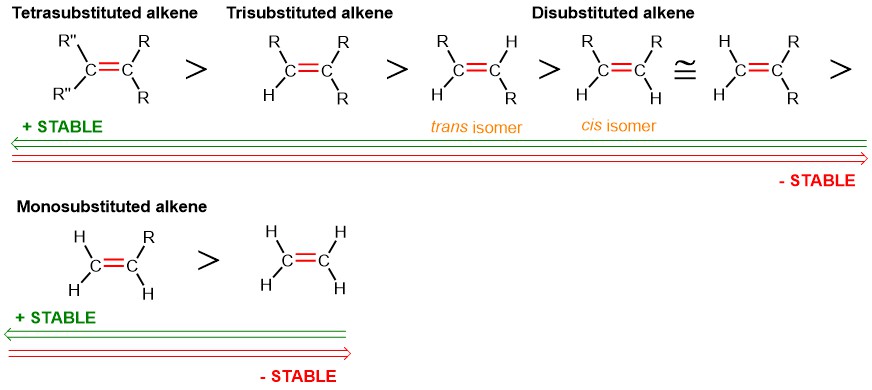
Therefore, in general, we can derive the following rule: the more substituted alkene is more stable than a less substituted one, and the trans alkene is more stable than the cis alkene.
But what is this trend due to? Let’s first look at why a more substituted alkene is more stable than an unsubstituted one.
This is due to a fundamental reason: the more substituted alkene has more alkyl R groups near the double bond, which results in the donation of electron density from the R groups (electron-rich and with sp³-hybridized carbons) to the sp²-hybridized carbons of the double bond, which are more electronegative than the sp³ carbons (see Figure 11).
This phenomenon, known as hyperconjugation, actually involves an overlap of molecular orbitals, which I will explain in more detail in a separate article. In any case, the explanation provided above is more than sufficient to answer a question on the subject thoroughly during an Organic Chemistry I exam.
It is also important to note that, despite the greater steric hindrance of more substituted alkenes, the hyperconjugative effect compensates so effectively for steric repulsions that a tetrasubstituted alkene—thus highly hindered—is significantly more stable than a monosubstituted alkene, where steric repulsions are minimal.
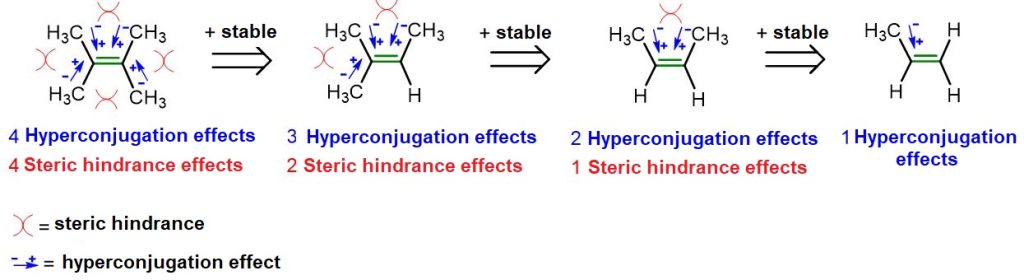
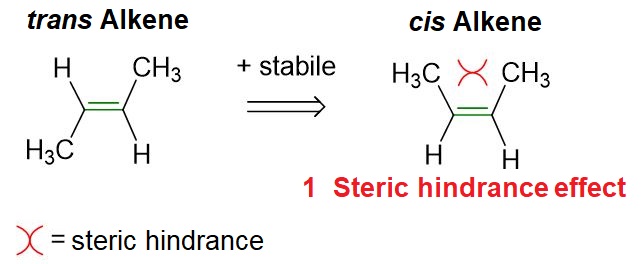
Let us conclude this paragraph by explaining the greater stability of the trans isomer compared to the cis isomer. In this context, the simplest and most widely accepted explanation is that, in the cis isomer, the two R groups are very close to each other and therefore tend to repel one another, thereby destabilizing the entire molecule. In contrast, in the trans isomer, the same R groups are farther apart and thus do not exhibit any repulsion.
Physical properties of alkenes
Alkenes have physical properties similar to those of alkanes: they are also nonpolar molecules and insoluble in water. They are soluble in solvents such as diethyl ether, alcohols, hexane, etc.
The first three alkenes (ethylene, propylene, and butene) are gases; the others, up to C₂₀H₄₀, are colorless liquids with densities lower than that of water. The higher alkenes, on the other hand, are solids.
Boiling points generally increase with chain length, but they also depend on the shape of the molecule and the type of isomerism. Several examples of this can be seen in Table 4. Comparing the boiling points of 1-alkenes, it is evident that, moving from ethylene with 2 carbon atoms to 1-decene with 10 carbon atoms, the boiling point rises dramatically, from -104°C for ethylene to 171°C for 1-decene.
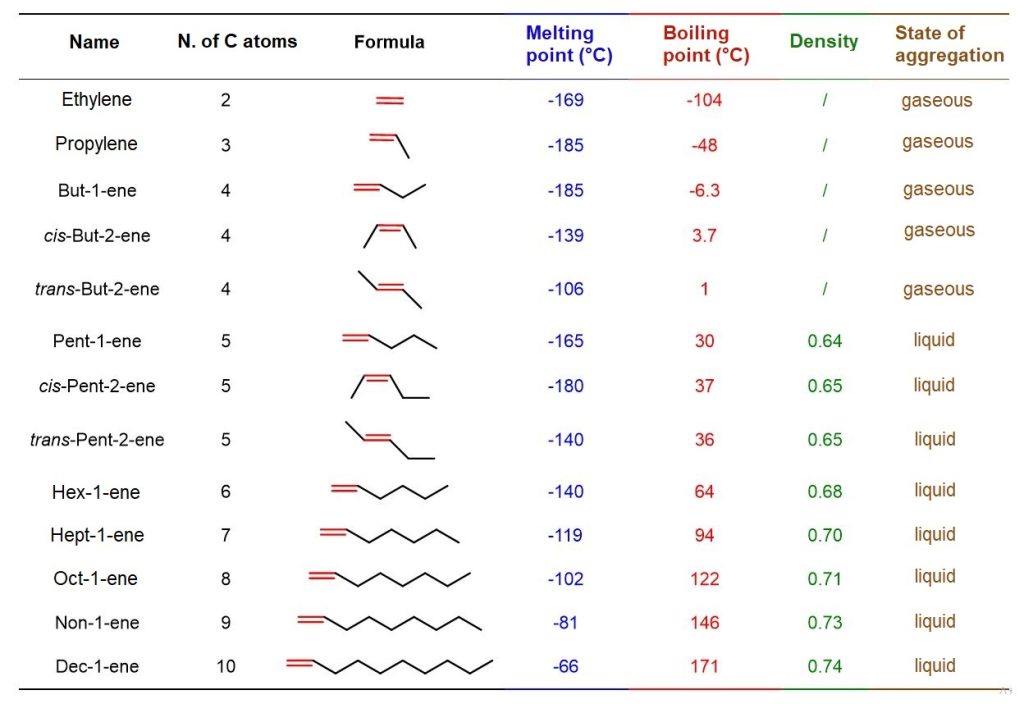
Regarding cis–trans isomerism, cis alkenes generally have higher boiling points than trans alkenes. This phenomenon occurs because trans alkenes are less polar than cis alkenes. In fact, the dipole moments generated by the hyperconjugative electron-donating effect of the R substituents to the sp² carbons of the double bond cancel out in the trans isomer, since the dipole vectors point in the same line but in opposite directions, making the overall polarity of the trans molecule zero. This does not happen in the cis isomer, where the dipole moments add up, resulting in a polar molecule. See an example in Figure 13.

Reactivity of alkenes
Alkenes mainly undergo two classes of reactions: electrophilic addition and polymerization. Both directly involve the double bond.
Let us examine both of them schematically, in order to obtain an overall picture.
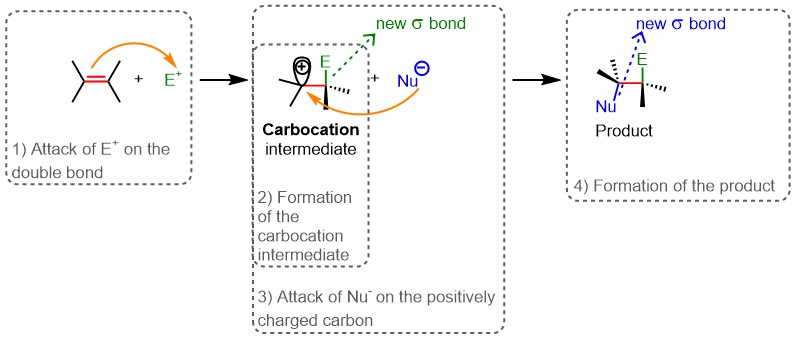
Electrophilic addition: an overview
The double bond is an electron-rich center; therefore, electrophilic molecules (electron-deficient species) are attracted to it. Electrophilic addition indeed involves an electrophile (E⁺) that attacks the double bond (Step 1, Figure 14). As a result, the double bond breaks and a reaction intermediate is formed, featuring a new σ bond between E⁺ and one of the carbon atoms involved in the double bond. This intermediate also carries a positive charge on the other carbon atom that did not react with E⁺, because that carbon now forms only three bonds instead of the four typical of its valence. The intermediate in question is therefore called a carbocation (Step 2, Figure 14). Subsequently, the carbocation reacts with a nucleophile (Nu, an electron-rich species), which, attracted by the positive charge, forms a σ bond with the positively charged carbon (Step 3, Figure 14). The reaction product is thus formed: a saturated molecule in which the double bond has been replaced by two new σ bonds (Step 4, Figure 14).
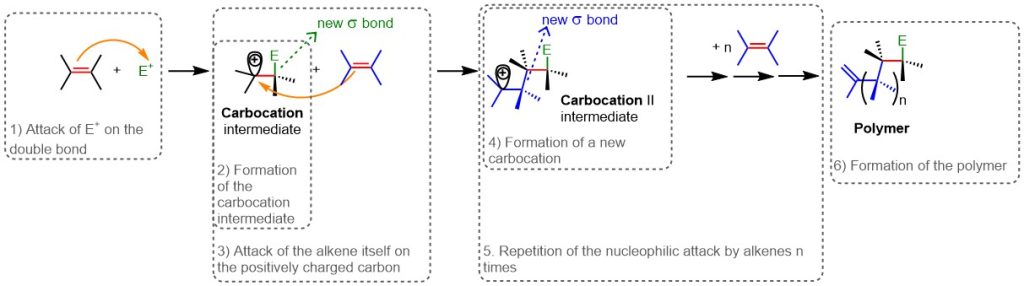
Polymerization
If no nucleophiles are present during the electrophilic addition reaction, the alkene itself—that has not yet reacted—can act as a nucleophile, leading to the polymerization of alkenes. This is possible because the double bond of alkenes is an electron-rich center and can, in fact, function as a nucleophile.
In more detail, after the attack of an electrophile on the double bond (Step 1, Figure 15) and the formation of the carbocation (Step 2, Figure 15), in polymerization another alkene molecule reacts with the carbocation (Step 3, Figure 15). This generates a new carbocation (Step 4, Figure 15), which can in turn react with another alkene molecule, and so on (Step 5, Figure 15). The process repeats many times, producing a polymer (Step 6, Figure 15)—a high-molecular-weight molecule in which molecular units repeat regularly along the length of the polymer chain.
Conclusion
In this article, I have provided you with a brief yet comprehensive overview of alkenes, covering their nomenclature, hybridization, cis–trans isomerism, stability, physical properties, and reactivity.
If all this information is overwhelming you, don’t worry! 💡
You can test what you’ve learned with our free quiz on alkenes, perfect for reviewing the material in an interactive and engaging way.
Do you prefer to study at your own pace? 📚
Download the PDF or DOCX file of this article and read it comfortably offline as well.
And to truly reinforce the concepts in your long-term memory, try our concept map on alkenes: a practical and effective way to memorize and summarize the material!⚗️
References
1) Tokue, I.; Fukuyama, T.; Kuchitsu, K. Molecular Structures of Propene and 3,3,3-Trifluoropropene Determined by Gas Electron Diffraction. Journal of Molecular Structure 1973, 17 (2), 207–223. https://doi.org/10.1016/0022-2860(73)85164-6.
2) Van Hemelrijk, D.; Van Den Enden, L.; Geise, H. J.; Sellers, H. L.; Schaefer, L. Structure Determination of 1-Butene by Gas Electron Diffraction, Microwave Spectroscopy, Molecular Mechanics, and Molecular Orbital Constrained Electron Diffraction. J. Am. Chem. Soc. 1980, 102 (7), 2189–2195. https://doi.org/10.1021/ja00527a007
3) Almenningen, A.; Anfinsen, I. M.; Haaland, A. Electron Diffraction Studies of cis– and trans-2-Butene. Acta Chem. Scand. 1970, 24, 43–49.
4) Ter Brake, J. H. M.; Mijlhoff, F. C. Trans-Pent-2-Ene. Electron Diffraction, Vibrational Analysis and Molecular Mechanics. Journal of Molecular Structure 1981, 77 (3–4), 253–263. https://doi.org/10.1016/0022-2860(81)80069-5.
5) Ter Brake, J. H. M. Cis-Pent-2-Ene. Electron Diffraction, Vibrational Analysis and Molecular Mechanics. Journal of Molecular Structure 1984, 118 (1–2), 63–71. https://doi.org/10.1016/0022-2860(84)85180-7.
This lesson

Alkenes-A step by step guide
€
4.00
Download as pdf (unchageable) file

Alkenes-A step by step guide
€
5.00
Downoald as docx (editable) file