The unmissable publication
The important publications that a chemist must know. Today we have chosen:
The Grignard reaction – Unravelling a chemical puzzle
Peltzer, R. M., Gauss, J., Eisenstein, O., & Cascella, M. (2020). Journal of the American Chemical Society, 142(6), 2984–2994.
‘’More than 100 years since its discovery, the mechanism of the Grignard reaction remains unresolved…The Grignard reaction puzzle is finally assembling together with potential impacts on related chemistry’’.

INTRODUCTION
In our previous article, we described the groundbreaking discovery of organomagnesium reagents, pioneered by the man you hated chemistry: Victor Grignard. However, even after a century since this discovery, the mechanism of the Grignard reaction has remained a puzzle for chemists worldwide.
Recently, Cascella’s group has shed light on this obscure and ancient topic, providing new insights to the scientific community through quantum chemical calculations and ab initio molecular dynamics. [1]
In this article, we aim to definitively clarify the mechanism of the Grignard reaction based on the most recent literature data.
STATE OF THE ART
‘’Victor Grignard. Photograph by GL Arlomer, Lyons’’ by Wellcome Collection [CC BY 4.0] via https://www.lookandlearn.com…
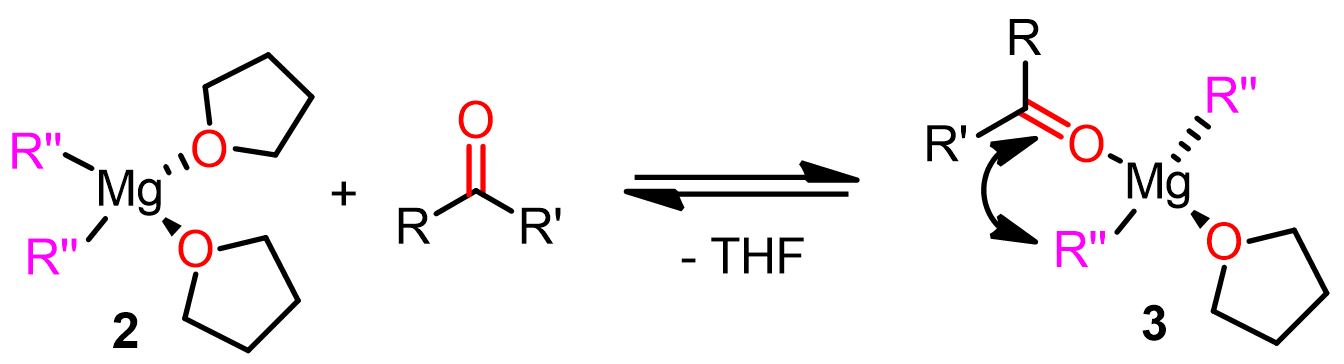
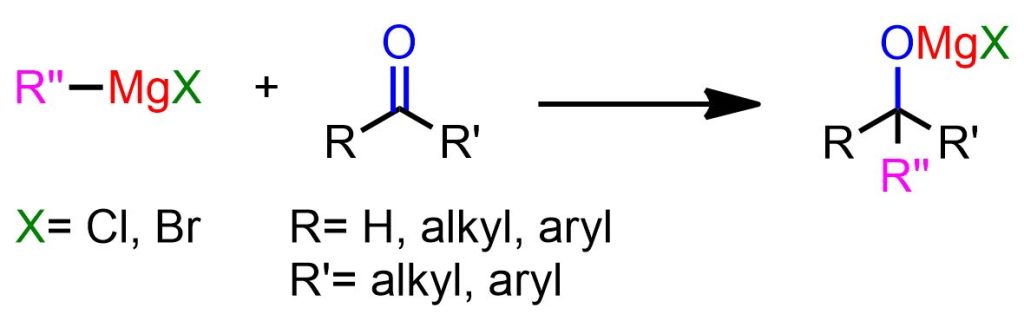
The Grignard reaction involves the reaction of organomagnesium compounds with an electrophile to form new C-C bonds. In this article, we will specifically investigate the reaction of RMgX with ketones and aldehydes (Figure 1) to afford substituted alcohols.
So far, two mechanisms have been suggested by chemists:
The radical mechanism (Figure 2a);
The nucleophilic addition (Figure 2b).
In previous literature, the electrophilic substrate seemed to be the discriminating factor between the two mechanisms. Specifically, nucleophilic addition was confirmed for aliphatic aldehydes, whereas the radical pathway was observed with substrates possessing low reduction potential, such as aromatic aldehydes and ketones.[2]
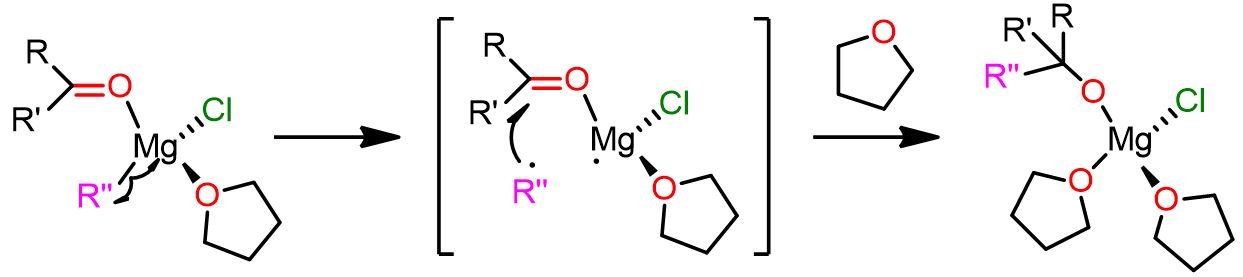
Figure 2a- Possible radical mechanism: homolytic Mg-C bond cleavage, followed by recombination of the radicals.

Figure 2b- Possible nucleophilic mechanism: heterolytic Mg-C bond cleavage, followed by nucleophilic addition to the carbonyl group.
The challenge in rationalizing the mechanism stems from two main factors:
– The empirical evidences of two types of mechanisms: radical or nucleophilic;
– The existence of complex mononuclear, dinuclear, and polynuclear organomagnesium complexes in solution.
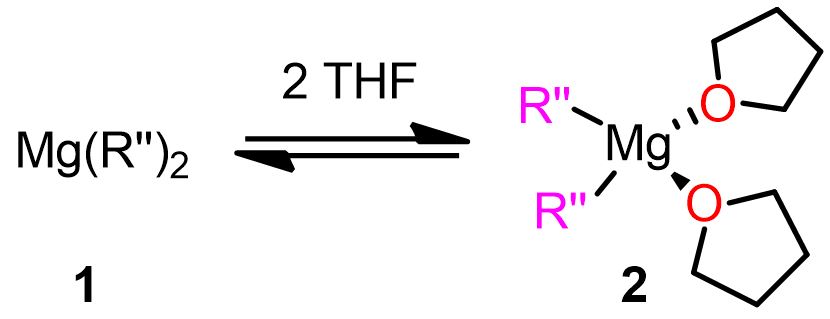
Regarding the last point, it is known that RMgX exists in solution in a variety of forms, depending on the solvent and concentration. However, the two most famous chemical species are those involved in the so-called Schlenk equilibrium (Figure 3), where monoalkyl magnesium compounds (RMgX) interconvert into dialkyl magnesium compounds (MgR2) by formation of dimers and oligomers. These Mg complexes can play a role in the mechanism of Grignard reaction.[3]
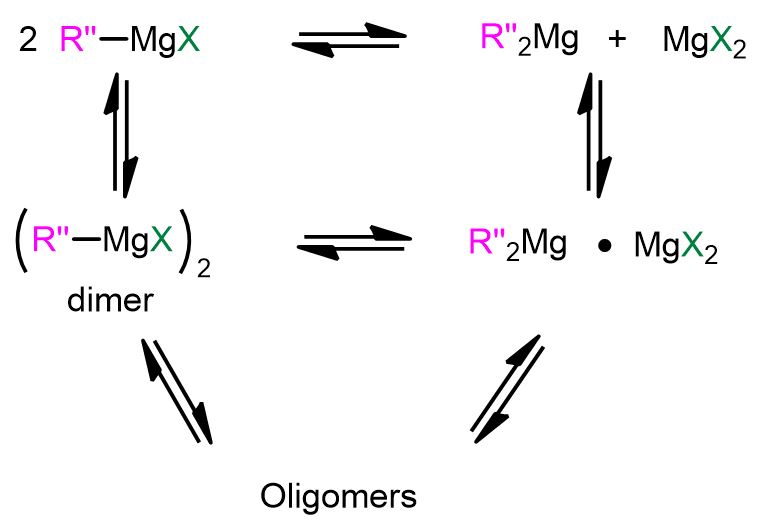
THE UNRAVELLED MECHANISM OF GRIGNARD REACTION
Among the known and confusing information about the Grignard mechanism, in 2020 Cascella’s group finally pieced together the puzzle and presented a convincing mechanism that explains all the previous experimental results. Through computational chemistry, they revealed the most likely pathways for this reaction.
The mechanism of Grignard reaction can occurr with both nucleophilic addition and radical pathway. However, the preferred mechanism is due to the nature of the electrophilic substrate:
Substrates with lower reduction potential and bulky substituents follow the radical mechanism.
Substrates with alkyl groups attached to the carbonyl moiety or those with less bulky substituents generally undergo nucleophilic addition.
In the following sections of this article, we will cover both mechanism in more detail.
The nucleophilic addition
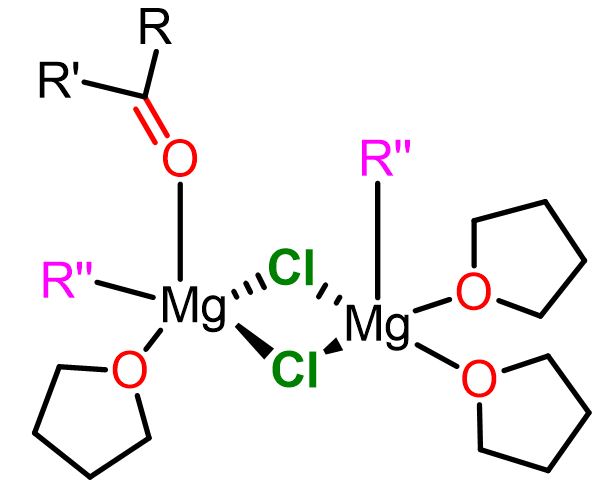
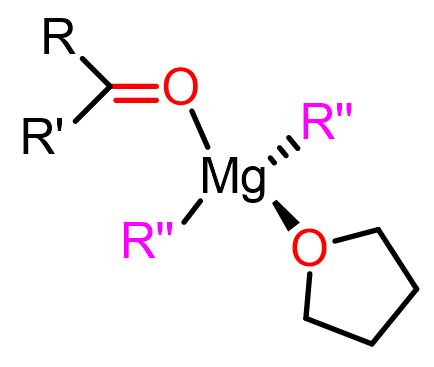
The Cascella’s team has calculated two major reactive magnesium complexes for the nucleophilic pathways. One is a mononuclear complex with the carbonyl substrate (O=CR2) and the alkyl nucleophile (R”) in a cis position (geminal position). The second reactive complex is a dinuclear one consisting of the carbonyl electrophile and the alkyl nucleophile bound to two different magnesium atoms (vicinal position). The two complexes are shown below.
Both of these complexes are reactive and contribute to the formation of the final product. The predominance of one complex over the other depends on the Schlenk equilibrium of that particular Grignard reagent.
Now, let’s explore the nucleophilic pathway with both of these complexes.
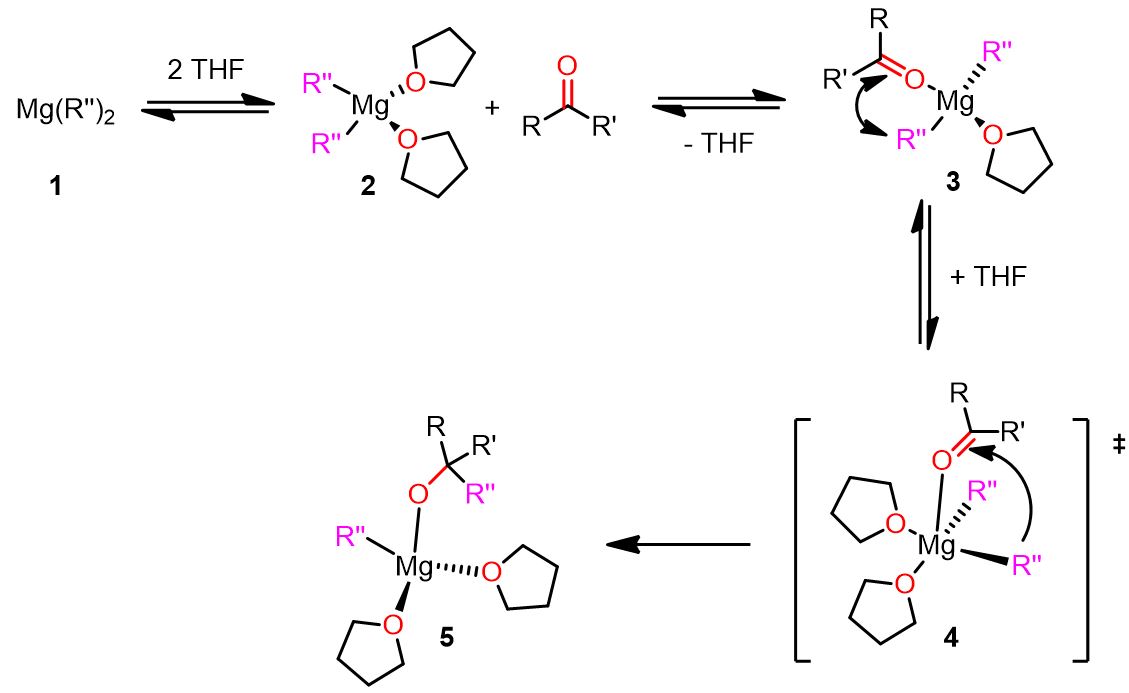
Polar mechanism with the geminal complex
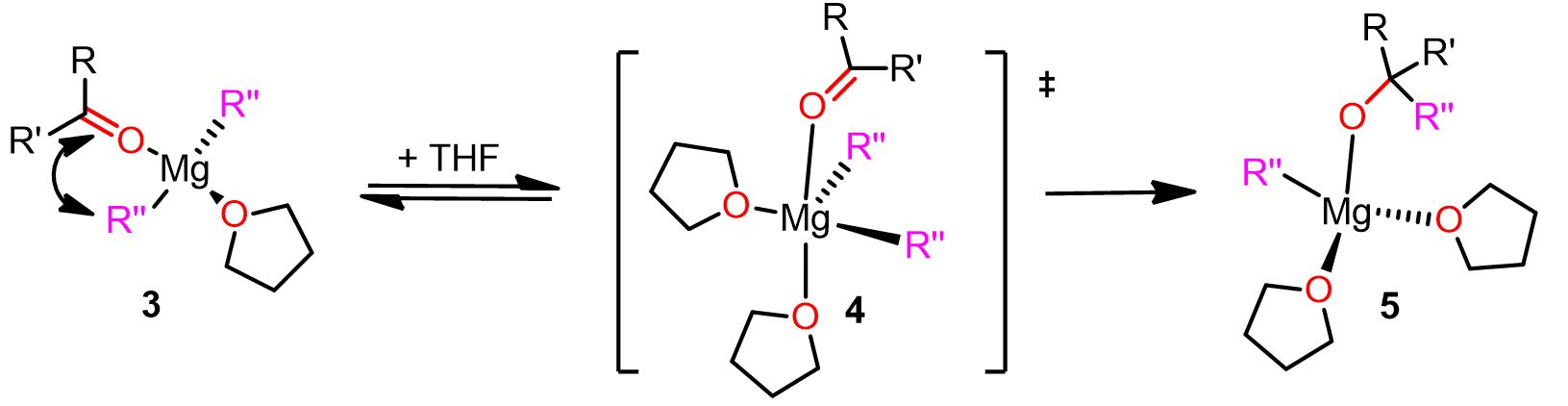
The geminal mechanism was named by Cascella’s team to refer to the geminal mononuclear reactive organomagnesium complex, which is one of the key chemical species formed during this pathway.
The mechanism is depicted below. Click on each step to see the entire pathway.
As you can see, in the above-described mechanism, the solvent molecules play a pivotal role in the reaction. For instance, in the last step, the entering THF molecule forces the carbonyl compound to go in the apical position and favors the cleavage of Mg-R” by donating electron density to the Mg.
Therefore, choosing the right solvent for the Grignard reaction can be important for the success of the reaction. Electron-donating solvents, indeed, make the magnesium more reactive and the nucleophile a better leaving group.
A complete illustration of the geminal mechanism is also displayed below.
Polar mechanism with the vicinal complex
This second competitive pathway for the Grignard reaction involves a dinuclear complex as reactive intermediate. In this complex, the two reacting species (R” and the carbonyl compound) are in apical positions and bound to two different Mg atoms (6 in Figure 5). The complex is also highly solvatated and this cause the electrophile and the nucleophile to be in closer proximity.
This second mechanism is depicted in the following picture.
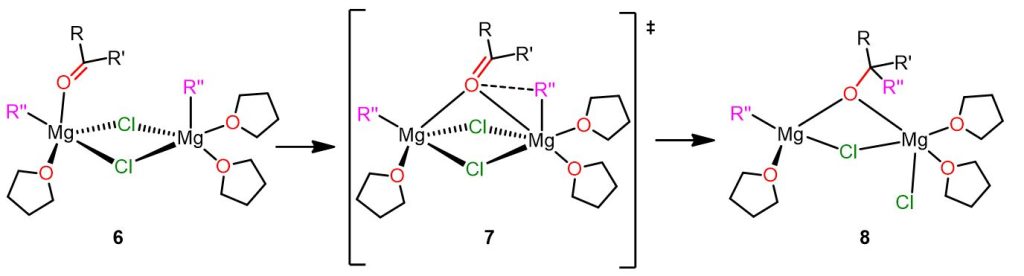
In this mechanism, specie 6 is formed during the Schlenk equilibrium and is one of the most abundant species. Moreover, this dinuclear complex also exhibits lower activation energy compared to the previous mononuclear complex 3.
Complex 6 undergoes rearrangement leading to transition state 7, where the electrophilic carbonyl compound bridges the two Mg atoms. In this transition state, the first Mg is pentacoordinated, whereas the second is octahedral. Now the carbonyl group is closer to the nucleophilic alkyl group R”, and a bond between them is formed. Moreover, the Cl atom, positioned opposite to the carbonyl compound, breaks its bond with the first Mg atom and becomes a terminal ligand.
In product 8, the first Mg atom is tetracoordinated, whereas the second is pentacoordinated. As you can see, the product bridges between the two Mg atoms.
The radical mechanism
The other competitive mechanism of Grignard reaction is the radical one. However, the homolytic cleavage of R-Mg bond has a high bond dissociation energy; therefote alkyl radical cannot be formed in pure solution of the Grignard reagents.
The formation of radicals, and indeed the radical mechanism, is only possible when the electrophilic substrate has low reduction potential. This suggests that it is the substrate that promotes the radical pathway.
Before understanding why low reduction potential substates can promote the radical mechanism, let’s see the radical mechanism in the picture below.
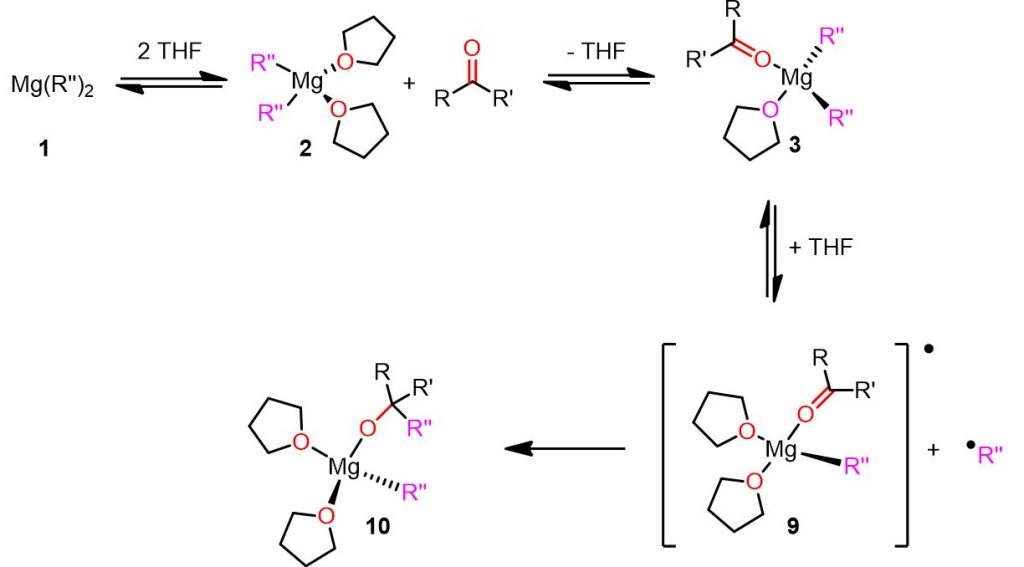
In the proposed radical mechanism, the steps from species 1 to 3 resemble the nucleophilic pathway (Figure 4) and involve the formation of a magnesium complex with the coordinated substrate (see complex 3). However, in the radical mechanism, one of the Mg-R” bonds undergoes homolytic cleavage, resulting in the formation of a radical magnesium complex 9 and a radical R” fragment. Subsequently, the radical R” fragment reacts with the electrophilic carbonyl group on complex 9 to yield species 10, with the alkoxide product bound to the Mg atom.
As we have already mentioned, the radical mechanism can only occur with substrates possessing low reduction potentials, such as aromatic carbonyl compounds like benzophenone and fluorene. Conversely, alkyl ketones and alkyl aldehydes will exclusively undergo the nucleophilic mechanism.
This occurs because the electron density of the unpaired complex 9 is primarily localized on the carbonyl group. Consequently, aromatic substrates can delocalize the unpaired electron within their structures through resonance, thereby reducing the bond dissociation energy of the Mg-R” bond and consequently lowering the energetic barrier for the formation of the radical species 9.
Additionally, substrates with low-lying π empty orbitals (indicative of low reduction potential) are often bulky. The significant bulkiness of these substrates impedes the nucleophilic pathway, wherein flexibility of the magnesium complexes is crucial for the reaction’s feasibility.
In conclusion, the radical pathway is favored by both electronic factors (such as substrates with low reduction potential) and steric considerations (in the case of bulky substrates).
CONCLUSION
After a century of debate, the mechanism of the Grignard reaction has finally been elucidated.
In particular, we have observed that two main pathways are viable: the radical and nucleophilic routes.
The radical mechanism manifests when the Grignard reaction is conducted with aromatic and bulky substrates. Aromaticity stabilizes the organomagnesium radical intermediate 9 through electron delocalization, whereas the steric hindrance hampers the nucleophilic route.
Conversely, the nucleophilic pathway occurs with alkyl aldehydes and alkyl ketones or with aromatic substrates that are not bulky, such as benzaldehyde.
The nucleophilic pathway can proceed through various routes, due to the formation of numerous organomagnesium compounds in solution. Nonetheless, the reaction mechanism adheres to the following primary principles:
The reaction occurs when both the substrate and the nucleophile are within the coordination sphere of Mg centers;
The number of Mg atoms in the reactive complex is not crucial, as both mononuclear and dinuclear complexes exhibit similar activation barriers. Likewise, the substrate and the nucleophile can reside on the same or nearby Mg atom (geminal or vicinal mechanism).
The solvent should be considered a reactant since it promotes the structural reorganization of the Mg reactive complexes.
References
1) Peltzer, R. M., Gauß, J., Eisenstein, O., & Cascella, M. (2020b). The Grignard reaction – unraveling a chemical puzzle. Journal of the American Chemical Society, 142(6), 2984–2994. https://doi.org/10.1021/jacs.9b11829.
2) Ashby, E. C., & Bowers, J. R. J. (1981). Organometallic reaction mechanisms. 17. Nature of alkyl transfer in reactions of Grignard reagents with ketones. Evidence for radical intermediates in the formation of 1,2-addition product involving tertiary and primary Grignard reagents. Journal of the American Chemical Society, 103(9), 2242–2250. https://doi.org/10.1021/ja00399a018.
3) Peltzer, R. M., Eisenstein, O., Nova, A., & Cascella, M. (2017). How solvent dynamics controls the Schlenk equilibrium of Grignard reagents: A computational study of CH3MgCl in tetrahydrofuran. the Journal of Physical Chemistry. B, 121(16), 4226–4237. https://doi.org/10.1021/acs.jpcb.7b02716